| Back Issues |
|
|
|
|
ACS Members can sign up to receive C&EN e-mail newsletter. |
|
|
 Join ACS |
| |||||||||
|
| |||||||||
|
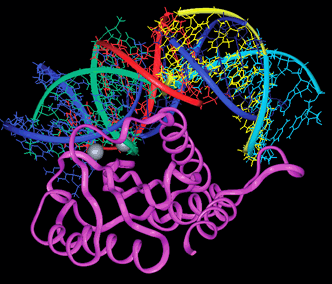
The first detailed molecular binding model consistent with experimental data on the complex of HIV integrase and viral DNA has been obtained by James M. Briggs of the University of Houston and coworkers [J. Med. Chem., published online Jan. 13, http://dx.doi.org/10.1021/jm0301890]. HIV integrase binds to a DNA copy of the virus's genome and catalyzes insertion of the DNA into a host chromosome, inducing the host cell to synthesize viral proteins. The enzyme is thus an essential part of the viral life cycle and a target for the design of therapeutic inhibitors. But the search for such agents has been impeded by the lack of any experimental integrase-DNA structure. The new study predicts a specific and plausible binding pattern in the integrase-DNA complex (shown)--information that should aid inhibitor design efforts. The researchers used an efficient parallel-docking method developed at Scripps Research Institute and the University of California, San Diego, to rapidly compute energies for more than 113 billion possible intermolecular configurations--"a procedure considered computationally intractable a few years ago," the researchers note. Nanofiber scaffold grows neurons A solid scaffold made of self-assembling nanofibers can selectively direct differentiation of neural progenitor cells, according to a new report [Science, published Jan. 22, http://www.sciencemag.org/cgi/content/abstract/1093783v1]. Samuel I. Stupp, professor of materials science, chemistry, and medicine at Northwestern University, and his colleagues direct the differentiation of neural progenitor cells using peptide amphiphiles that self-assemble into nanofibers in the presence of cells or tissue. The nanofibers have peptide sequences (epitopes) on their surface that are known to promote and direct the growth of dendrites and axons. Neural progenitor cells that are encapsulated in the gel-like matrix formed by the aggregation of these fibers rapidly differentiate into neurons, with about 35% testing positive for a neuron marker protein after only one day. In contrast, the researchers observe little formation of astrocytes, another type of neural cell, after seven days. By mixing nanofibers containing the active epitopes with others that don't, Stupp and his colleagues find that the epitope density plays a major role in the observed differentiation. Self-assembly with helical proteins Researchers report a method for creating a new class of supramolecules and fibers out of dendrimers and leucine zippers, a common and important helical protein structural motif. The strategy, developed by assistant chemistry professor Indraneel Ghosh and colleagues at the University of Arizona, could be used to create numerous varieties of biomaterials and noncovalent complexes, such as protein inhibitors or enzymatic complexes. The group synthesized two different macromolecules, each composed of a core dendrimer to which they covalently attached four leucine zipper peptides [J. Am. Chem. Soc., 126, 734 (2004)]. They then mixed each macromolecule with individual peptides, which assembled noncovalently into complexes. Ghosh's group also reports that the two macromolecules, when mixed together, self-assemble into helical fibers. Metal ion biosensor Few good methods exist for selective detection of metal ions. Yet microorganisms detect metal ions selectively with the help of the MerR protein family. Chuan He, an assistant professor of chemistry at the University of Chicago, and graduate student Peng Chen used the MerR specificity to design a strategy for creating selective, fluorescent metal ion biosensors [J. Am. Chem. Soc., 126, 728 (2004)]. MerR proteins bind to specific sequences of double-stranded DNA. When a specific metal ion then binds to the protein, the protein untwists the DNA helix and breaks apart two base pairs in the middle of the duplex. If a fluorescent cytosine analog, pyrrolo-C, is incorporated into just the right spot on the DNA strand, it fluoresces after being unpaired. Chen and He built two examples of what they think can be a sensor using any MerR protein. They used E. coli's MerR, which binds primarily to Hg2+, and CueR, which responds to Cu+, Ag+, or Au+. Addition of the metal ion typically caused a threefold increase in fluorescence in seconds. In addition to metal sensing, He believes that the sensors can be used to probe the behavior of newly discovered MerR proteins. Déjà vu for carbon cycling Black carbon, a relatively inert material made up of polyaromatic hydrocarbons and amorphous carbon, originates from plants burned incompletely during wildfires and from fossil-fuel combustion. A study led by chemistry graduate student Angela F. Dickens of the University of Washington, Seattle, has now determined that a fraction of this carbon appears to loop through Earth's carbon cycle over geological timescales, a finding that could affect assumptions made in climate change and other studies [Nature, 427, 336 (2004)]. The researchers used mass spectrometry to measure the amount of 13C and 14C in black carbon and codeposited bulk organic carbon in ocean and terrestrial core samples. From preindustrial core sections, the team chemically isolated and quantified a completely inert portion of black carbon that is old enough to have lost its 14C signature. The 13C data for this fraction mimics that of fossilized organic matter, leading the researchers to conclude that about half of the black carbon in some sediments originates from the weathering of rocks, where it likely formed during metamorphosis of kerogen (shale oil), rather than from direct combustion of biomass, as originally thought. The "radiocarbon dead" fraction also could affect 14C dating in general, making samples appear older than they are. | |||||||||
|
Chemical &
Engineering News | |||||||||
|
| ||||||||
| ||||||||